If you double the average speed of the molecules in a gas, by what factor does the pressure change?12/15/2023  What would happen to the pressure if the number of particles in the container increases and the temperature remains the same? The pressure comes from the collisions of the particles with the container. Pressure is proportional to temperature, if the number of particles and the volume of the container are constant. The force exerted by the particles per unit of area on the container is the pressure, so as the temperature increases the pressure must also increase. The proportionality constant is (2/3)R and R is the gas constant with a value of 0.08206 L atm K -1 mol -1 or 8.3145 J K -1 mol -1.Īs the temperature increases, the average kinetic energy increases as does the velocity of the gas particles hitting the walls of the container. 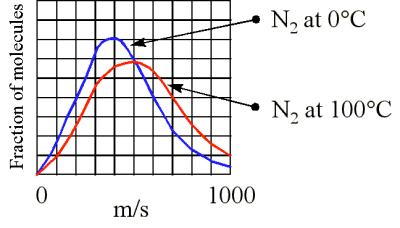  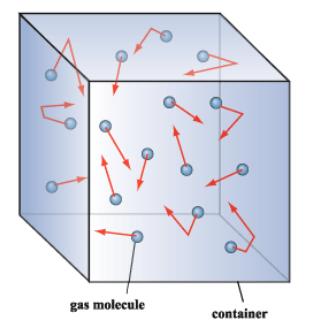 
We know that temperature is proportional to the average kinetic energy of a sample of gas.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
